Health state transition models
Added on 01/07/2022
Always wondered how health economic models work? Good news: this blog is developed to guide you through the basic elements of ‘health state transition models’.
‘Simulation models’ are an important instrument to simulate the impact of the introduction of innovation over time. These models are commonly used in health economic assessment to predict the potential costs over time taking into account the clinical efficacy.
If the concerned innovation would be introduced on national scale, it would be very difficult to simulate each single patient over time. Although we can rely on supercomputers driven by artificial intelligence, it would be very difficult to predict the potential outcome accurately. In addition, the time needed to analyse the design of the ‘ideal model’ and the efforts to construct and validate the model would not be in balance with the targeted result. More so, models built to obtain market access are ‘single use’ models. Therefore a simplification of the ‘ideal model’ is recommended. Of course, the uncertainty of the predicted outcome will increase but this is outside the scope of this blog.
An experienced health economic modeller will understand the needs for which the model should serve. If the concerned innovation will impact the short term outcome (impact over a few days), it doesn’t make sense to extend the time horizon over several decades. And when a model is designed over a long term horizon (chronic diseases) it would be unlogic to program the model per day. The ideal time horizon should be selected in a way to fully cover the financial and clinical impact of the innovation.
All models will analyse the impact of a certain variable (diagnostic test, e-health application, new pharmaceutical formulation, …) versus the target population. For example: for advanced therapy medicinal products (ATMPs) like gene therapies it will be important to understand the patient characteristics and the impact the targeted genetic disorders (life threatening, disability, …) as well as the innovation (ATMPs) can have on the patients. Therefore, the model should include at least the variables which can impact the costs over time as well the patients’ quality of life. It’s up to the modeller to design the model in sufficient detail to enable simulation of the effect of the innovation and the disease while reducing complexity as much as possible.
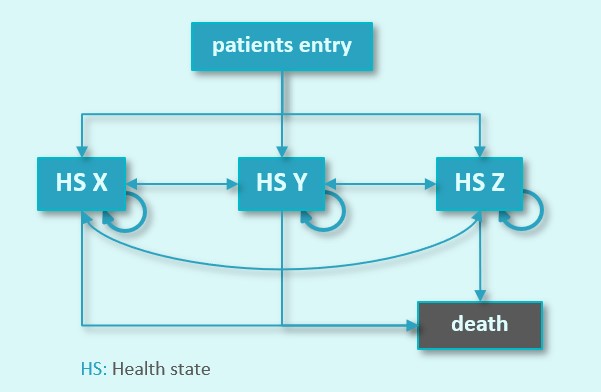
The methodology used is to predefine a number of health states which are representative of the disease pattern in the target population. All patients will enter the model in a certain health state; a proportion of these will switch (or not) towards another health state. The new technology may have an impact on the proportion of patients that will shift over the predefined health states and/or the time needed before patients will shift. The more health states in the model, the more the patients will be distributed. When a life time horizon is selected, all patients will enter in the mortality health state (some patients will die due to the disease, while others will die due to other causes).
Some models are designed as such that patients can move towards another health state but never return back towards the former health state. These models are defined as ‘partitioned health state transition models’. Oncology related models are a typical example of these. Patients progress over time and once progressed, the patient will switch from one health state towards the progressed health state, and eventually will be adsorbed in the mortality health state.
Another type of model are the ‘Markov models’. Markov models allow to simulate patients in 2 directions. They can move over several health states and remain in the concerned health state, return to a former health state and switch towards a new health state. These models are typically used to simulate patients with chronic diseases or to simulate the impact of innovation over a longer time horizon.
If a model is designed to simulate patient transitions per month, the patients will stay in the concerned health state or move towards another health state when a new model cycle starts. When built in Excel, each line represents 1 model cycle and the columns represent the health states. At start of the model, all patients (100%) will enter the model in cycle 1 (upper row) in the same health state (1 cell). The next model cycle (row 2) will redistribute the 100% of the patients towards the potential health states (columns). This continues over as many model cycles as defined.
The sum of all proportions in 1 row will always count up to 100%. The sum of all proportions in the columns will inform the user about the percentage of time a patient stays in a certain health state over the time horizon of the model.
Each column represents a health state and the cost of a health state is defined per model cycle. Example: if the cost of 1 month in the concerned health state is 1.000 € and 25% of the patients stay in this health state, the cost would be 250 € in the concerned model cycle. The same calculation can be made for all patients distributed over the other health states.
This way, the overall cost per model cycle can be calculated (sum of all columns in 1 line) and the cost per health state over the model horizon can be calculated as well (sum of 1 column). When summing up all costs from all health states the overall cost per average patient is known.
The same methodology can be used for the patients’ ‘Quality of Life’ expressed in utilities (we refer to our blog: ‘Quality of Life – be vigilant!’ for more information on utilities).
Finally, the model will generate at least two outcomes: one for the concerned innovation in the scope and one for the standard of care (current clinical practice). These two results can be subtracted from each other to calculate the Incremental Cost Effectiveness Ratio (ICER).
Assuming that the time horizon and the granularity of the model (model cycles and health states) are well defined, a model is nothing without reliable clinical data. In general, the clinical data are extracted from clinical studies. Of course, the model should be designed as such that the available clinical data can be used correctly without the need for complex conversions and/or extrapolations. But, this is for a different blog.
Our message with this blog is to motivate all people with health economic interests to think about the design and to align with the needs of your innovation while avoiding model complexity. In the past few years, we saw the building of expensive (fancy) models to tell simple stories. We think that at least in some cases a more simplified approach would have resulted in similar success towards market access.
Feel free to contact us in case you prefer to work the simplified way without losing the value story to bring your innovation to the market!
We are happy to welcome you to provide more details and insights.